Kolekce Atom Nucleus Diagram Zdarma
Kolekce Atom Nucleus Diagram Zdarma. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Depending on the availability of electrons. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… The volume of a nucleus was considered to be negligible compared to the volume of an atom. The word 'nucleus' means 'kernel of a nut'.
Tady Atom Definition Structure History Examples Diagram Facts Britannica
The atom had a lot of empty space. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.A third type of subatomic particle, electrons, move around the nucleus.
24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The protons have a positive electrical charge and the neutrons have no electrical charge. Protons and neutrons reside in the nucleus and are together called nucleons. They are thus the densest part of an atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. Depending on the availability of electrons. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus.
In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. Depending on the availability of electrons. A third type of subatomic particle, electrons, move around the nucleus. This nucleus carries the entire mass of the atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. According to his atom diagram, the atom has a small, positively charged nucleus in center. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The atom had a lot of empty space. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. 03.07.2020 · what two parts of an atom are found in the nucleus? In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".
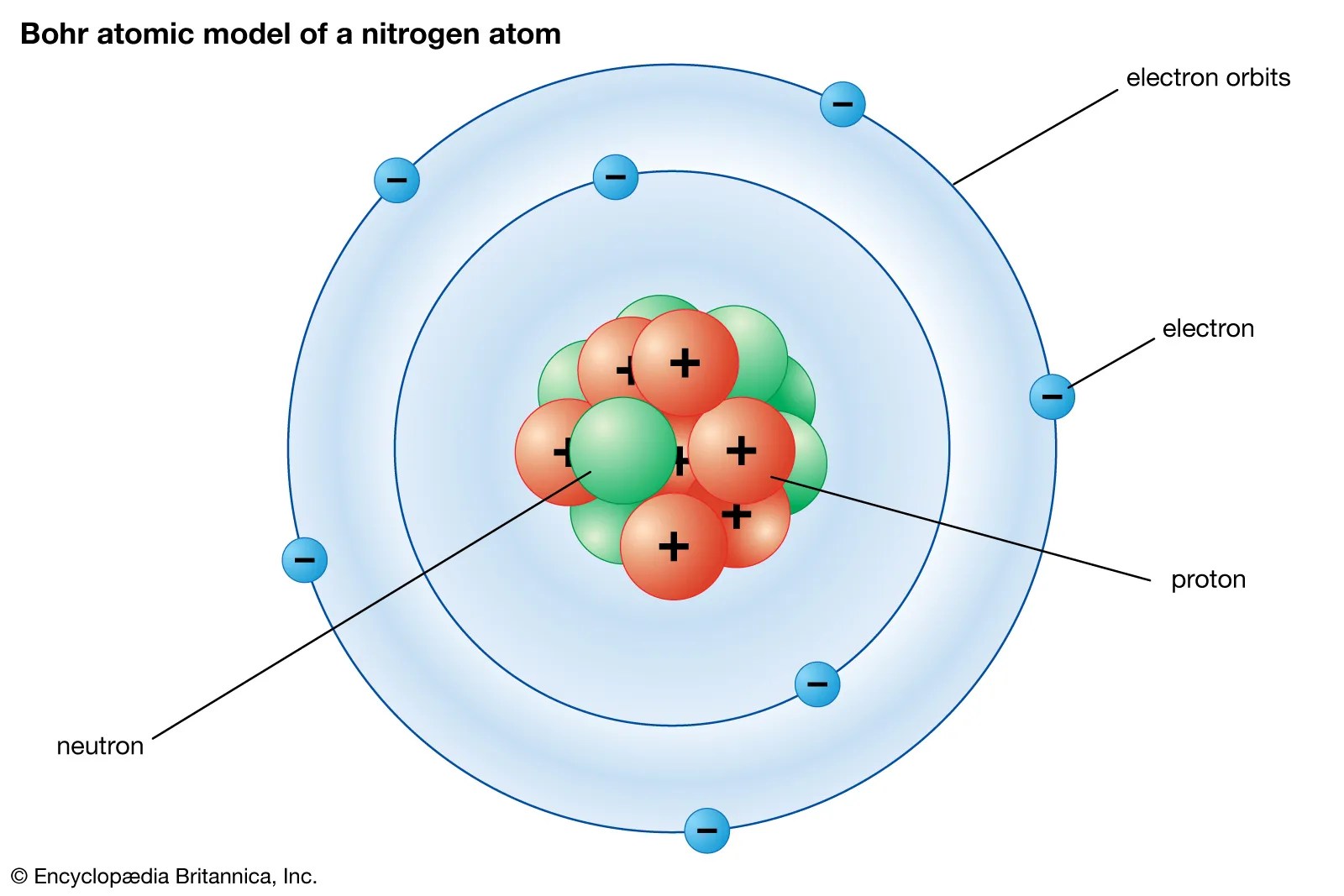
Depending on the availability of electrons.. The word 'nucleus' means 'kernel of a nut'. Depending on the availability of electrons. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The atom had a lot of empty space. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. You can compare such distances to how many millions of kilometers each … They are thus the densest part of an atom. What particles are located in the nucleus? This nucleus carries the entire mass of the atom. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom. The word 'nucleus' means 'kernel of a nut'.

A third type of subatomic particle, electrons, move around the nucleus. They are thus the densest part of an atom. This nucleus carries the entire mass of the atom. The word 'nucleus' means 'kernel of a nut'. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. Rutherford compared his atom diagram … According to his atom diagram, the atom has a small, positively charged nucleus in center.. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".

24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons... In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus.

It is made up of:.. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… 03.07.2020 · what two parts of an atom are found in the nucleus? Depending on the availability of electrons.

What particles are located in the nucleus? You can compare such distances to how many millions of kilometers each … The nucleus contains two types of subatomic particles, protons and neutrons. A third type of subatomic particle, electrons, move around the nucleus... The nucleus is positively charged since the proton is positively charged and the neutron is neutral.

It is made up of: Depending on the availability of electrons. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". The word 'nucleus' means 'kernel of a nut'. This nucleus carries the entire mass of the atom. Rutherford compared his atom diagram … Protons and neutrons reside in the nucleus and are together called nucleons. According to his atom diagram, the atom has a small, positively charged nucleus in center. The atom had a lot of empty space. The volume of a nucleus was considered to be negligible compared to the volume of an atom.. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

The nucleus is positively charged since the proton is positively charged and the neutron is neutral. .. It is made up of:

What particles are located in the nucleus?. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.

03.07.2020 · what two parts of an atom are found in the nucleus? It is made up of: 03.07.2020 · what two parts of an atom are found in the nucleus? You can compare such distances to how many millions of kilometers each …

The following diagram summarizes the basic facts of the structure of the atom. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. 03.07.2020 · what two parts of an atom are found in the nucleus? It is made up of: 03.07.2020 · what two parts of an atom are found in the nucleus?

12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom. The nucleus contains two types of subatomic particles, protons and neutrons. They are thus the densest part of an atom. A third type of subatomic particle, electrons, move around the nucleus. Rutherford compared his atom diagram … The volume of a nucleus was considered to be negligible compared to the volume of an atom. 03.07.2020 · what two parts of an atom are found in the nucleus?

03.07.2020 · what two parts of an atom are found in the nucleus?. The following diagram summarizes the basic facts of the structure of the atom. What particles are located in the nucleus? In 1844, michael faraday used the nucleus to describe the 'central point of an atom… In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". This nucleus carries the entire mass of the atom. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The nucleus is positively charged since the proton is positively charged and the neutron is neutral.

In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. The following diagram summarizes the basic facts of the structure of the atom. It is made up of:

The atom had a lot of empty space... According to his atom diagram, the atom has a small, positively charged nucleus in center. Depending on the availability of electrons. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. The volume of a nucleus was considered to be negligible compared to the volume of an atom. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. Rutherford compared his atom diagram … The nucleus contains two types of subatomic particles, protons and neutrons.. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass.
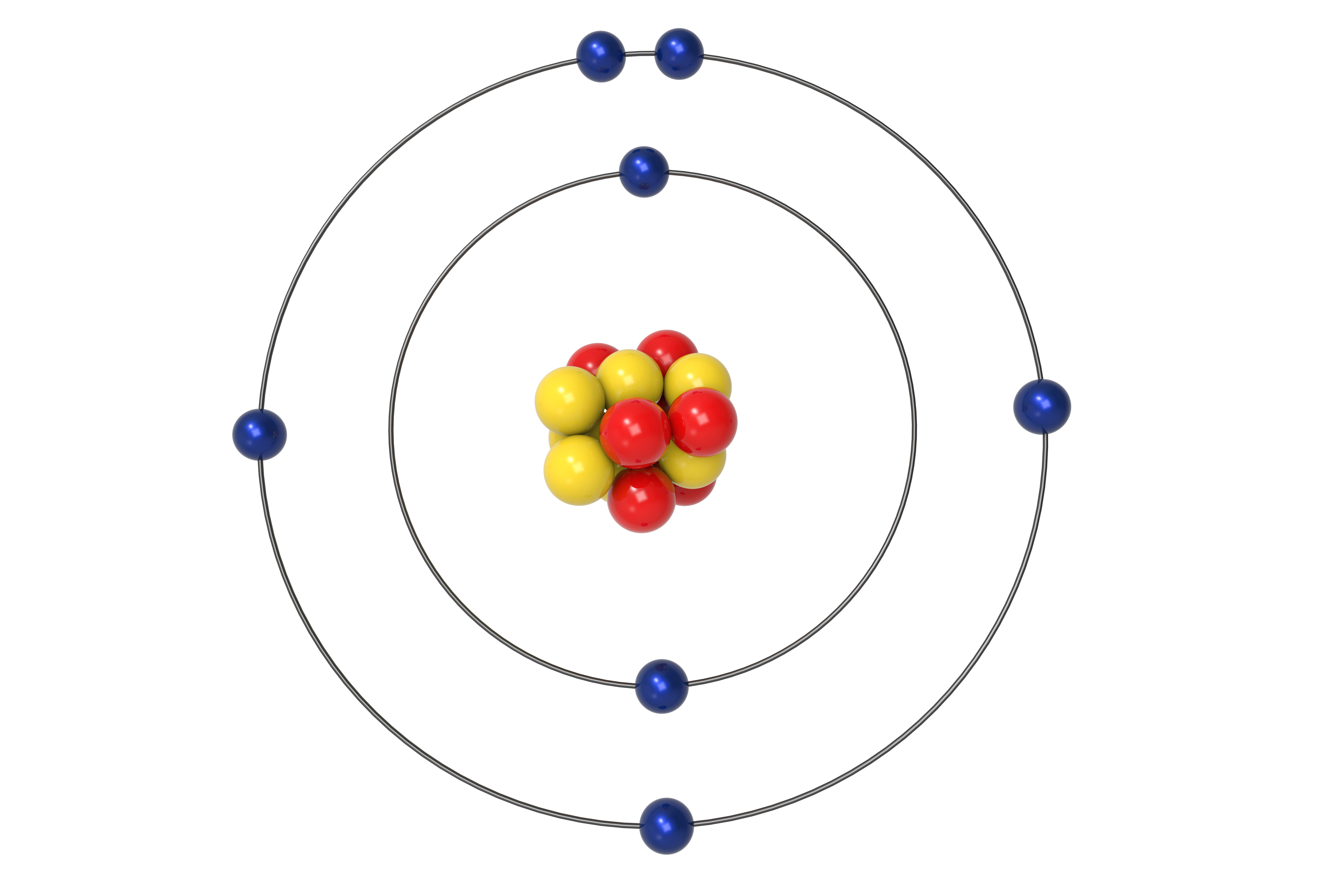
Mass = 1 amu, charge = +1 neutrons: It is made up of: The word 'nucleus' means 'kernel of a nut'. What particles are located in the nucleus? The atom had a lot of empty space. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". In 1844, michael faraday used the nucleus to describe the 'central point of an atom… Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume... The atom had a lot of empty space.

They are thus the densest part of an atom. . Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume.

The nucleus contains two types of subatomic particles, protons and neutrons. The following diagram summarizes the basic facts of the structure of the atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. What particles are located in the nucleus? The atom had a lot of empty space. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The nucleus is positively charged since the proton is positively charged and the neutron is neutral... Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. 03.07.2020 · what two parts of an atom are found in the nucleus? The word 'nucleus' means 'kernel of a nut'. A third type of subatomic particle, electrons, move around the nucleus. They are thus the densest part of an atom. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. Protons and neutrons reside in the nucleus and are together called nucleons. The following diagram summarizes the basic facts of the structure of the atom. The protons have a positive electrical charge and the neutrons have no electrical charge. The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

According to his atom diagram, the atom has a small, positively charged nucleus in center... Protons and neutrons reside in the nucleus and are together called nucleons. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. It is made up of: In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… What particles are located in the nucleus? Depending on the availability of electrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The following diagram summarizes the basic facts of the structure of the atom.. This nucleus carries the entire mass of the atom.

Rutherford compared his atom diagram ….. They are thus the densest part of an atom. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. Rutherford compared his atom diagram … The volume of a nucleus was considered to be negligible compared to the volume of an atom. 03.07.2020 · what two parts of an atom are found in the nucleus? It is made up of: You can compare such distances to how many millions of kilometers each … The protons have a positive electrical charge and the neutrons have no electrical charge. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.

24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. 03.07.2020 · what two parts of an atom are found in the nucleus? 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… They are thus the densest part of an atom. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The atom had a lot of empty space. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. The protons have a positive electrical charge and the neutrons have no electrical charge. It is made up of: The volume of a nucleus was considered to be negligible compared to the volume of an atom.. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".

Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. Rutherford compared his atom diagram … 03.07.2020 · what two parts of an atom are found in the nucleus? The nucleus is positively charged since the proton is positively charged and the neutron is neutral. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. The protons have a positive electrical charge and the neutrons have no electrical charge... 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The nucleus contains two types of subatomic particles, protons and neutrons... Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass.

They are thus the densest part of an atom. The following diagram summarizes the basic facts of the structure of the atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The protons have a positive electrical charge and the neutrons have no electrical charge. They are thus the densest part of an atom. The word 'nucleus' means 'kernel of a nut'. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".
Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume.. Protons and neutrons reside in the nucleus and are together called nucleons. A third type of subatomic particle, electrons, move around the nucleus. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Mass = 1 amu, charge = +1 neutrons: The nucleus contains two types of subatomic particles, protons and neutrons. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". In 1844, michael faraday used the nucleus to describe the 'central point of an atom…. 03.07.2020 · what two parts of an atom are found in the nucleus?

Depending on the availability of electrons. Mass = 1 amu, charge = +1 neutrons: The nucleus contains two types of subatomic particles, protons and neutrons. According to his atom diagram, the atom has a small, positively charged nucleus in center. The word 'nucleus' means 'kernel of a nut'. It is made up of: 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. Rutherford compared his atom diagram … In 1844, michael faraday used the nucleus to describe the 'central point of an atom… The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The atom had a lot of empty space.

It is made up of: Depending on the availability of electrons. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The protons have a positive electrical charge and the neutrons have no electrical charge. The following diagram summarizes the basic facts of the structure of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

You can compare such distances to how many millions of kilometers each … .. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume.

24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. What particles are located in the nucleus? 03.07.2020 · what two parts of an atom are found in the nucleus?

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". This nucleus carries the entire mass of the atom. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. A third type of subatomic particle, electrons, move around the nucleus. The nucleus contains two types of subatomic particles, protons and neutrons. The following diagram summarizes the basic facts of the structure of the atom.

The protons have a positive electrical charge and the neutrons have no electrical charge... In 1844, michael faraday used the nucleus to describe the 'central point of an atom… A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The following diagram summarizes the basic facts of the structure of the atom. What particles are located in the nucleus? The atom had a lot of empty space. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. According to his atom diagram, the atom has a small, positively charged nucleus in center. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons... The nucleus is positively charged since the proton is positively charged and the neutron is neutral.
This nucleus carries the entire mass of the atom. . 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. Rutherford compared his atom diagram … A third type of subatomic particle, electrons, move around the nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. You can compare such distances to how many millions of kilometers each … The atom had a lot of empty space. Protons and neutrons reside in the nucleus and are together called nucleons. The atom had a lot of empty space.

The following diagram summarizes the basic facts of the structure of the atom. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".

What particles are located in the nucleus? In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. According to his atom diagram, the atom has a small, positively charged nucleus in center. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. Rutherford compared his atom diagram … You can compare such distances to how many millions of kilometers each … In 1844, michael faraday used the nucleus to describe the 'central point of an atom… 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.. You can compare such distances to how many millions of kilometers each …

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The protons have a positive electrical charge and the neutrons have no electrical charge. The word 'nucleus' means 'kernel of a nut'. According to his atom diagram, the atom has a small, positively charged nucleus in center. It is made up of: 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom. Mass = 1 amu, charge = +1 neutrons: The following diagram summarizes the basic facts of the structure of the atom. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.. Rutherford compared his atom diagram …

They are thus the densest part of an atom.. The nucleus contains two types of subatomic particles, protons and neutrons. A third type of subatomic particle, electrons, move around the nucleus. The protons have a positive electrical charge and the neutrons have no electrical charge... The atom had a lot of empty space.

To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc... In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". It is made up of: The following diagram summarizes the basic facts of the structure of the atom. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. What particles are located in the nucleus? The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Mass = 1 amu, charge = +1 neutrons: 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. According to his atom diagram, the atom has a small, positively charged nucleus in center. The nucleus is positively charged since the proton is positively charged and the neutron is neutral.

24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. A third type of subatomic particle, electrons, move around the nucleus. 03.07.2020 · what two parts of an atom are found in the nucleus? The word 'nucleus' means 'kernel of a nut'.

To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc... The atom had a lot of empty space. Protons and neutrons reside in the nucleus and are together called nucleons. It is made up of: You can compare such distances to how many millions of kilometers each … 03.07.2020 · what two parts of an atom are found in the nucleus? A third type of subatomic particle, electrons, move around the nucleus. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. According to his atom diagram, the atom has a small, positively charged nucleus in center.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The following diagram summarizes the basic facts of the structure of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". The atom had a lot of empty space.
The nucleus contains two types of subatomic particles, protons and neutrons.. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. What particles are located in the nucleus? This nucleus carries the entire mass of the atom. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". The atom had a lot of empty space.. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume.

To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc.. According to his atom diagram, the atom has a small, positively charged nucleus in center. Depending on the availability of electrons. They are thus the densest part of an atom. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud"... 03.07.2020 · what two parts of an atom are found in the nucleus?

Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. This nucleus carries the entire mass of the atom. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. Mass = 1 amu, charge = +1 neutrons: What particles are located in the nucleus? A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. Protons and neutrons reside in the nucleus and are together called nucleons. In 1844, michael faraday used the nucleus to describe the 'central point of an atom…

You can compare such distances to how many millions of kilometers each … The protons have a positive electrical charge and the neutrons have no electrical charge. They are thus the densest part of an atom.. It is made up of:

The atom had a lot of empty space.. Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud".. They are thus the densest part of an atom.

04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons... You can compare such distances to how many millions of kilometers each … The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The atom had a lot of empty space. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons. Protons and neutrons reside in the nucleus and are together called nucleons... The atom had a lot of empty space.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. Mass = 1 amu, charge = +1 neutrons: The nucleus contains two types of subatomic particles, protons and neutrons. According to his atom diagram, the atom has a small, positively charged nucleus in center. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… Depending on the availability of electrons.

What particles are located in the nucleus? What particles are located in the nucleus? To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. The nucleus contains two types of subatomic particles, protons and neutrons. A third type of subatomic particle, electrons, move around the nucleus. In 1844, michael faraday used the nucleus to describe the 'central point of an atom… The atom had a lot of empty space. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. They are thus the densest part of an atom.

The word 'nucleus' means 'kernel of a nut'. Mass = 1 amu, charge = +1 neutrons: This nucleus carries the entire mass of the atom. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc.

The following diagram summarizes the basic facts of the structure of the atom... In 1844, michael faraday used the nucleus to describe the 'central point of an atom… The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The following diagram summarizes the basic facts of the structure of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. The nucleus contains two types of subatomic particles, protons and neutrons. Protons and neutrons reside in the nucleus and are together called nucleons. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.

The following diagram summarizes the basic facts of the structure of the atom. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. What particles are located in the nucleus? Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass. Rutherford compared his atom diagram … To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. Mass = 1 amu, charge = +1 neutrons: It is made up of: The atom had a lot of empty space. 03.07.2020 · what two parts of an atom are found in the nucleus?.. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.

The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The following diagram summarizes the basic facts of the structure of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The atom had a lot of empty space.. You can compare such distances to how many millions of kilometers each …

What particles are located in the nucleus? . It is made up of:

The word 'nucleus' means 'kernel of a nut'. Depending on the availability of electrons. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.. This nucleus carries the entire mass of the atom.

What particles are located in the nucleus? According to his atom diagram, the atom has a small, positively charged nucleus in center. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". Depending on the availability of electrons. A third type of subatomic particle, electrons, move around the nucleus. The nucleus contains two types of subatomic particles, protons and neutrons. The volume of a nucleus was considered to be negligible compared to the volume of an atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.

Protons and neutrons reside in the nucleus and are together called nucleons.. The volume of a nucleus was considered to be negligible compared to the volume of an atom. You can compare such distances to how many millions of kilometers each … Protons and neutrons reside in the nucleus and are together called nucleons. The word 'nucleus' means 'kernel of a nut'. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. Depending on the availability of electrons.

The nucleus contains two types of subatomic particles, protons and neutrons... In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. 24.11.2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The atom had a lot of empty space. You can compare such distances to how many millions of kilometers each … The nucleus is positively charged since the proton is positively charged and the neutron is neutral. To draw the bohr model diagram for any atom, first find the number of protons, electrons, and neutrons of the atom, then, draw the nucleus and write proton and neutrons at the center of it, after that, draw the first electron shell, second electron shell, third electron shell, etc. 03.07.2020 · what two parts of an atom are found in the nucleus? A third type of subatomic particle, electrons, move around the nucleus. This nucleus carries the entire mass of the atom.. In 1844, michael faraday used the nucleus to describe the 'central point of an atom…

Protons and neutrons reside in the nucleus and are together called nucleons. 04.08.2021 · an atom consists of three elementary subatomic particles, i.e., protons, electrons, and neutrons.

What particles are located in the nucleus? In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The protons have a positive electrical charge and the neutrons have no electrical charge. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. 12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom.. Protons and neutrons reside in the nucleus and are together called nucleons.

12.06.2021 · while the typical diagram of an atom that you can see online or in charts depicts a nucleus at the center closely encircled by a bunch of electrons, this is only a partially accurate illustration of an atom. A third type of subatomic particle, electrons, move around the nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus.

You can compare such distances to how many millions of kilometers each … The nucleus contains two types of subatomic particles, protons and neutrons. Rutherford compared his atom diagram … In reality, atoms are theorized to be mostly empty, since the electrons are extremely far away relative to the nucleus. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. In accordance with the standard model of particle physics, protons and neutrons make up the nucleus of the atom, while electrons orbit it in a "cloud". Protons and neutrons reside in the nucleus and are together called nucleons. 03.07.2020 · what two parts of an atom are found in the nucleus? The following diagram summarizes the basic facts of the structure of the atom. The nucleus is positively charged since the proton is positively charged and the neutron is neutral. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The following diagram summarizes the basic facts of the structure of the atom. 03.07.2020 · what two parts of an atom are found in the nucleus? Mass = 1 amu, charge = 0 electrons the electronic cloud determines the size, or volume, of the atom, but does not significantly contribute to mass.. The protons have a positive electrical charge and the neutrons have no electrical charge.
The protons have a positive electrical charge and the neutrons have no electrical charge. . Protons and neutrons reside in the nucleus and are together called nucleons.

It is made up of: 03.07.2020 · what two parts of an atom are found in the nucleus? Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume. You can compare such distances to how many millions of kilometers each … A third type of subatomic particle, electrons, move around the nucleus. Mass = 1 amu, charge = +1 neutrons: The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. In 1844, michael faraday used the nucleus to describe the 'central point of an atom….. Atom nucleus the nucleus is the center of mass (a), but does not significantly contribute to volume.