Kolekce Quantum Mechanical Model Of Atom Schrodinger Equation
Kolekce Quantum Mechanical Model Of Atom Schrodinger Equation. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation.
Tady Atomic Structure And Quantum Numbers Powerpoint Slides
Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.It is the quantum mechanical model of the atom that started from the schrödiger equation.
Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Until that time, electrons were only considered to rotate in circular orbits.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.
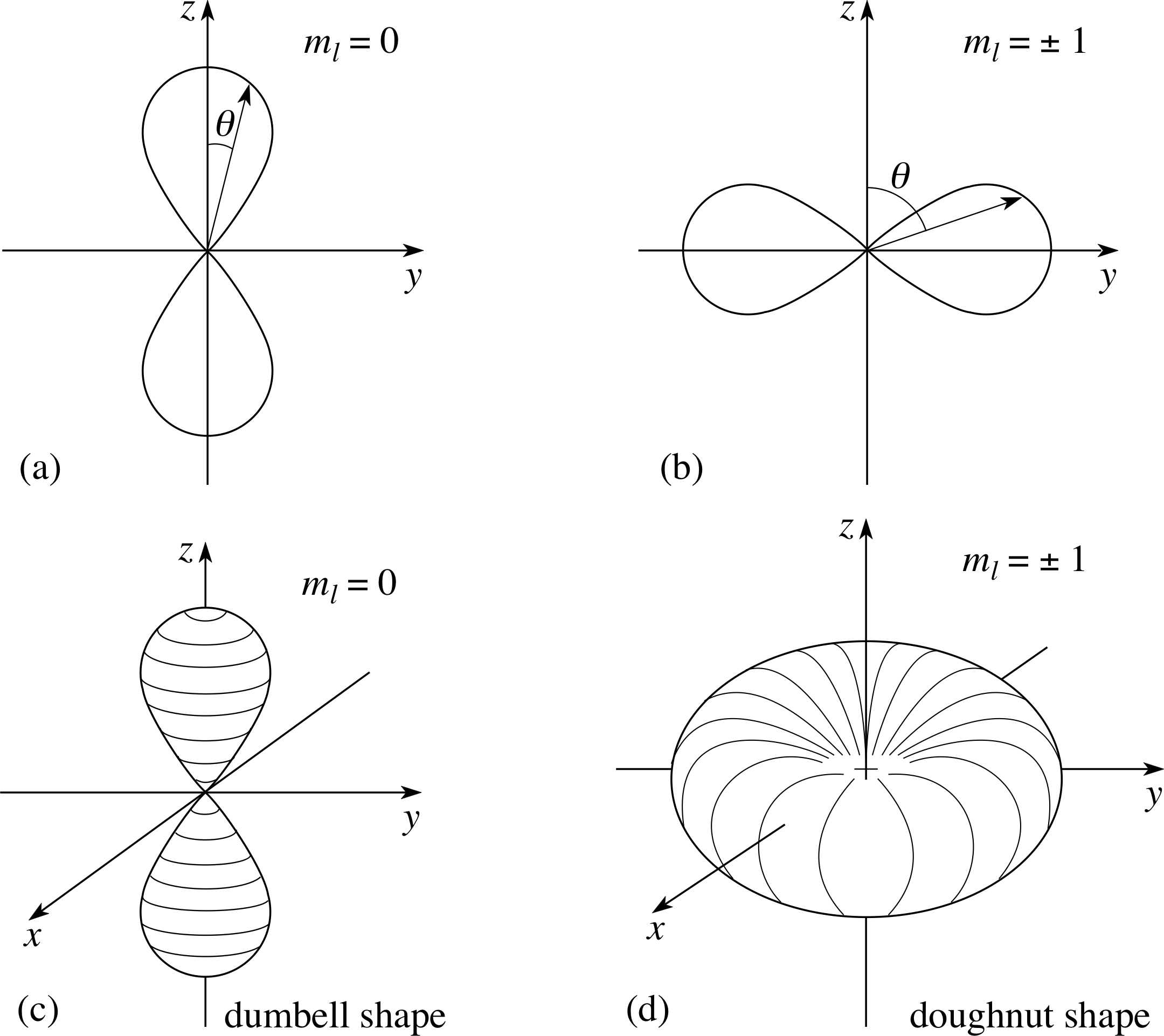
In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Until that time, electrons were only considered to rotate in circular orbits.. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. It is the quantum mechanical model of the atom that started from the schrödiger equation.

It is the quantum mechanical model of the atom that started from the schrödiger equation.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation... It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. It is the quantum mechanical model of the atom that started from the schrödiger equation.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation.

It is the quantum mechanical model of the atom that started from the schrödiger equation... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Until that time, electrons were only considered to rotate in circular orbits.

It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.
Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits.. Until that time, electrons were only considered to rotate in circular orbits.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation.

It is the quantum mechanical model of the atom that started from the schrödiger equation.. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.
It is the quantum mechanical model of the atom that started from the schrödiger equation... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Until that time, electrons were only considered to rotate in circular orbits.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. .. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

It is the quantum mechanical model of the atom that started from the schrödiger equation.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Until that time, electrons were only considered to rotate in circular orbits.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Until that time, electrons were only considered to rotate in circular orbits. . Until that time, electrons were only considered to rotate in circular orbits.

It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.
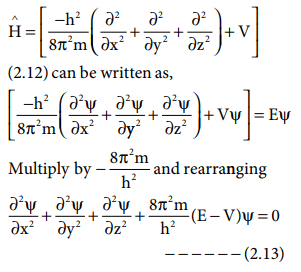
In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits... Until that time, electrons were only considered to rotate in circular orbits.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation.

Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.
Until that time, electrons were only considered to rotate in circular orbits... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits... Until that time, electrons were only considered to rotate in circular orbits.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

It is the quantum mechanical model of the atom that started from the schrödiger equation... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Until that time, electrons were only considered to rotate in circular orbits.
In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. It is the quantum mechanical model of the atom that started from the schrödiger equation.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... . In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

It is the quantum mechanical model of the atom that started from the schrödiger equation... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

Until that time, electrons were only considered to rotate in circular orbits.. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits.

Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.
Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. .. Until that time, electrons were only considered to rotate in circular orbits.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

It is the quantum mechanical model of the atom that started from the schrödiger equation. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,.

Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,... Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits. It is the quantum mechanical model of the atom that started from the schrödiger equation.

Until that time, electrons were only considered to rotate in circular orbits. Until that time, electrons were only considered to rotate in circular orbits. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

Until that time, electrons were only considered to rotate in circular orbits... It is the quantum mechanical model of the atom that started from the schrödiger equation. Until that time, electrons were only considered to rotate in circular orbits.. Until that time, electrons were only considered to rotate in circular orbits.
In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. Until that time, electrons were only considered to rotate in circular orbits.. Until that time, electrons were only considered to rotate in circular orbits.

Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation.. It is the quantum mechanical model of the atom that started from the schrödiger equation.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. It is the quantum mechanical model of the atom that started from the schrödiger equation. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. Until that time, electrons were only considered to rotate in circular orbits... In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.

In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom. Until that time, electrons were only considered to rotate in circular orbits. Erwin schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves. It is the quantum mechanical model of the atom that started from the schrödiger equation. Schrödinger's equation, , can be solved to yield a series of wave function , each of which is associated with an electron binding energy,. In 1926, erwin schrödinger developed this equation to determine the probability of finding an electron at a certain point in an atom.